SHARE
Innovative Medical Devices: The Future of Healthcare
Frede Jensen, the winner of the ICU Ventilators for COVID19: Design and Manufacturing Challenge, who submitted the design for the OxyVita emergency ventilator, shares with us his views on the future of healthcare and innovative medical devices. Frede is a British engineer with more than 30 years of experience in the development of medical devices and ventilators.
This week a Freeview TV broadcaster replayed the 1992 European Cup football semi-final between the Netherlands and Denmark, and I watched. The visual experience and graphical match information felt primitive today. There was no Video Assisted Referee (VAR), but this didn’t actually result in any serious decision errors. Importantly, the fundamental function of the game – the standard of football play – would still win world-class matches today. Something that has changed of course is the amount of money we now pay for watching ‘similar’ 2020 football. When in the year 2050 we take a historic view, then I can easily imagine that medical devices of 2020 will look primitive. But, will the differences really be that significant?
Just as in football, the traditional medical devices of 1990 are remarkably similar in function to those in 2020. For example, today’s lung ventilator displays better graphical representations of what is happening with the lung; but the methods and modes of ventilation are still those that were developed in the 1950s to 1970s. It demonstrates how it is easier to advance the technology for measuring and monitoring patients (or football) than it is to advance the technology that does something to the patient. We can define ‘value’ as the ratio between benefits and price. This has a nonlinear relationship, which differs at the economy and luxury ends of the market. Investment in more benefits invariably reaches a point where the higher rate of price increase will reduce value. This point of diminishing return tends to happen somewhere between the entry-level and mid-range price point. As a generalized rule, in the mid-range, for every +10% of added benefits, we can expect about +30% of the added price. 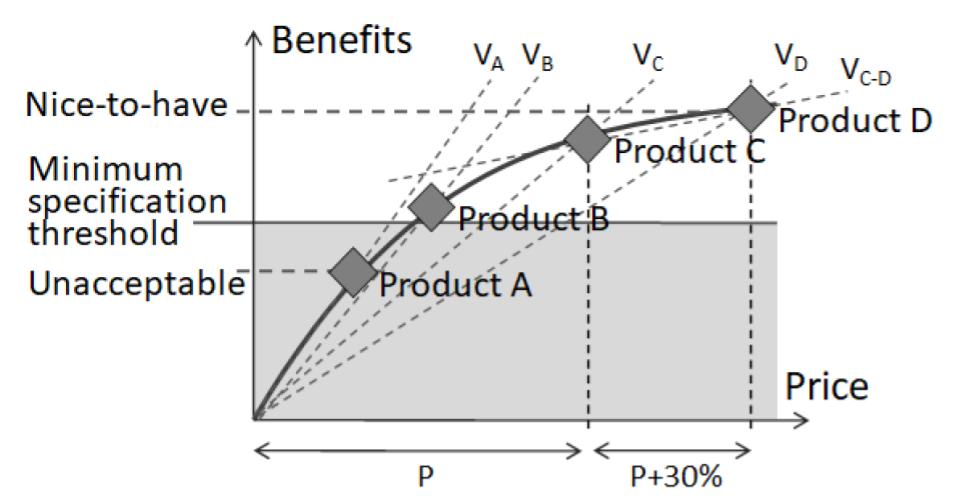 Value lines and the ‘natural law of pricing’ curve
Value lines and the ‘natural law of pricing’ curve
Covid-19’s overwhelming patient numbers exemplify how value-based equipment decisions can make a positive difference to public health outcomes. A hospital’s affordability-constrained purchasing choice could be between, say, 5 premium ventilators, 10 mid-range ventilators, or 15 entry-level ventilators – all of which essentially deliver the same therapy. The premium ventilators have features that require advanced infrastructure and expert user training, which are hard to establish and maintain. Ennomotive’s OxyVita is an example solution that is purposed for mass-supply urgency, without straining the healthcare budgets, and being just right for the lower levels of infrastructure and clinical skills that exist within disaster hospitals – while it still delivers an advanced lung protective volume mode of ventilation to patients.  OxyVita emergency ventilator
OxyVita emergency ventilator
Health is, of course, priceless. Clinicians and patients will always want the perceivably very best innovative medical devices, regardless of costs or operating difficulties. The argument that less can be more is therefore unwinnable. It does not need to be this way though, because manufacturing and distribution have in fact never been more efficient. Although being functionally very similar, there has been a significant technical evolution in innovative medical devices. Take, for example, the component costs for a lung ventilator, which have more than halved over the last 2 decades. The cost of a single-board computer that drives a touchscreen user interface has reduced from 400€ to just 50€ over this time, while performance has increased many folds.
Microprocessors and System-on-Chip solutions have become increasingly powerful and now integrate the features that previously required 10’s of more components, at 10 times the cost and size. Reduced components count – with fewer solder joints – and reduced reliance on analog signal conditioning have significantly improved reliability and accuracy. It is similar for cost reduction in plastic injection molding tools. Easily accessible 3D printing has made development prototyping quicker and cheaper than ever. What has increased significantly in costs, however, are the activities and time-to-market delays associated with regulatory conformity assessment procedures.
Innovative medical devices: 3 new trends
Drug-device combinations (DDCs)
These devices merge two – or three if we count biologic – fields of medicines. The transdermal (nicotine) patch is an example that has been around for decades, but this was kind of it for a long while. DDCs also include connected devices, such as a smart inhaler that connects to a mobile app for recording the rate and dosages of use. DDCs are in process of moving on to the next evolutionary stage, now that the enabling regulatory framework has been established. Implantable blood glucose sensors connected with servo-controlled insulin pumps are already common in diabetes management, for example. The Medtronic MiniMedTM range, manufactured by IntriCon Inc, is a current forefront example. In 2019, a Bigfoot Biomedical and Eli Lilly partnership announced they will improve the insulin dosing, by building artificial intelligence into the pump. This development is still at a concept testing stage. 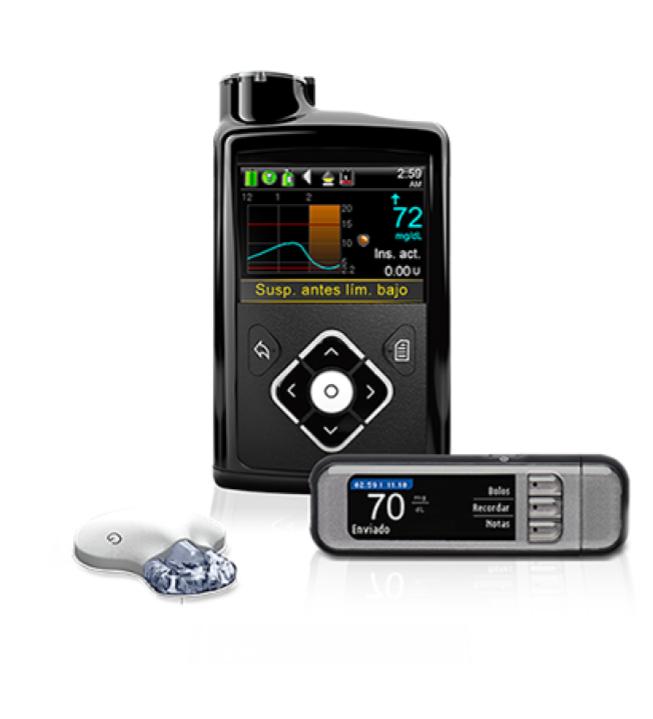
Image by MedTronic
There are huge numbers of yet unexploited healthcare applications for DDCs delivering better-targeted medicines. DDCs can enable drugs with severe side-effects to be delivered in smaller targeted doses. This includes new and highly promising drugs that could not previously be used. The technology promises to make lives better for millions of patients. According to one market research study, the DDCs market is growing at 8% per year and could reach about 30% of the world’s US$600 billion medical devices spend by 2024.
Preventive and predictive healthcare
A freshly published study into the “proactive, reactive and personalized use of technology to support aging populations in their own homes” in Spain – a global leader in telecare – in collaboration with the equipment manufacturer Tunstall, found that technology, including fixed in-home and wearable sensors combined with alerting devices, reduced emergency calls by 54% and ambulance mobilizations by 36%. Helping older people stay independent and self-sufficient at home for longer improves lives and frees up care funding. Using both individual biometric and lifestyle data, processed by artificial intelligence (AI) and combined with big data from across a wider population, will help predict or early detect the onset of health conditions – before they develop into acute or irreversible ill-health.
The Google Health app team, like many others, are investing heavily in “studying the use of artificial intelligence to assist in diagnosing cancer, predicting patient outcomes, preventing blindness, and much more” – all through a mobile app. It is easy to see how the spread of Covid-19 could have been better controlled if more proactive detection and tracking had been available earlier. The big issue is of course data security and privacy. Some markers for diseases are found in our DNA, for example, which none of us are keen on being held in a public database for widespread investigations. People must remain in control of their own data, while simultaneously allowing anonymized data to be gathered to support community research. It possibly also requires a shift away from healthcare being owned by big public health institutions, which are minimally funded and therefore must focus mainly on acute care.
Their preventive efforts tend to revolve around public health information campaigns. By definition, such institutions find it difficult to operate predictively on an individual basis – and nor would we like them to. Lastly, prevention and prediction can merge with fitness. “If exercise was on prescription, then it would be classed as a wonder drug,” says the UK Department for Health. The primary aim of fitness devices is no longer just about improving looks or sporting competitiveness. 20 minutes brisk walk is not just good for physical and mental health, but it can enable biometric sensors to evaluate key health parameters and detecting any trends. Devices such as Fitbits are already evolving in this direction, although yet at a primitive level. 
Image by Fitbit
Self-care, self-management
Innovative medical devices for this market are important in 3 ways:
- The WHO reports a current 7 million shortfall in healthcare professionals, which is set to reach 18 million by 2050. Self-care technologies can partly substitute for this shortfall.
- Patient involvement and motivation, from self-care, can in many situations deliver better outcomes.
- Dignity and pride from human self-sufficiency.
The number of available condition management apps is already in the thousands, ranging from simple pill reminders to mHealth integration with biometric sensors. Mental health is presently the most widespread application. Diabetes management is also high in the app's ranking. The devices can provide both acute and chronic disease support. As an example potential area, urinary tract infections (UTI), and falls are the most common causes for elderly people ending up in hospital – and hitting a downwards health slope. UTIs generally result from the insufficient fluid intake. Body temperature from the UTI and dehydration result in dizziness, which happens to be a leading cause of falls. The interrelated stages of events could be easily detected and reversed before they result in hospitalization.
A few years back, I managed a telehealth operation, which equipped patients to measure their own biometrics daily. The system uploaded the data via the phone to a remote monitoring station, which would test for certain trending parameters and alert to any changes. This intended design actually proved to be the system’s lesser valuable benefit, I think. People with respiratory diseases, for example, started to test their blood oxygen saturation before deciding to go to the shop. This prevented incidents of exacerbation, where they would have ended up in an ambulance. Patients would first-hand see the health benefits from exercise and medication, which motivated them to better manage their condition. Participating doctors reported that patients would show up with their data and tell the doctor how they are doing. Prior, it was the doctor doing the telling, which often felt meaningless to the individual and made them less likely to comply with the doctor’s prescription.
One thing that telehealth, or mHealth, does not resolve, however, is loneliness. A good proportion of our patients would not upload their daily data until an operator telephoned to remind them. This was not all due to forgetfulness. Sadly, many patients felt a need to use the service as an opportunity for a daily dose of social contact. Medical equipment-wise, Quanta Dialysis Technologies have now enabled home kidney dialysis. This procedure traditionally involves a complicated weekly round trip to a hospital clinic, where the dialysis procedure is performed over 3 hours. The relatively rapid changes in blood fluids and pressure that incur over 3 hours, when combined with the underlying disease, regularly results in cramps, nausea, and vomiting. The discomfort causes some patients to give up dialysis. About 80% of patients are capable of self-administering the procedure at home, where they can extend the dialysis time to 8 hours – or possibly even over-night while at sleep. This longer time significantly reduces the discomforting side-effects. It also eliminates the need for the person with compromised health having to attend a hospital clinic and potentially coming into contact with sources of infections. 
Image by Quanta Dialysis Technologies
New self-care solutions are also emerging in the under-served hearing care market. Nuheara’s IQbuds2 MAX now blurs the line between the medical device and consumer product. The Bluetooth headset features hearing aid functionality, incorporating hearing testing and the medical NAL-NL2 prescription formula for matching the amplification profile to the individual hearing loss. Similarly, Apple has in recent weeks announced they are about to upgrade their Airpod Pro with hearing aid functionality. This is a very strong beginning, and affordable too. Both the Nuhara and Apple devices can be bought over-the-counter for about 250€ per pair and they can effectively address 90% of all hearing impairment. What they have not yet fully developed, in my view, is the assurance of a good self-fit amplification profile and ‘acclimatization’ support; but both of these are easily fixable and will undoubtedly be rolled into future apps.  Image by Nuhara
Image by Nuhara
When self-care and self-management are linked to consumer devices, as already seen with Nuheara and Fitbit, it removes the unwanted ‘patient’ label and turns the user into ordinary health ‘consumer’. The self-caring person can always fall back on a telemedicine app, such as ‘Doctor on Demand’, for a video or virtual doctor consultation and recommendation. There are already 10’s of telemedicine apps available, which provide around the clock and on weekends doctor services when advice is needed. Importantly, they are on-demand and the user remains in control.  Image by Doctor on Demand
Image by Doctor on Demand
In summary, there is promising potential for making our 2020 approach to innovative medical devices look laughably primitive by 2050. I hope this will happen.
If you want to read more about innovative medical devices and other innovations, check out ennomotive's blog and discover what ennomotive can do for you. Join the engineering community